捲膜式薄膜
Alfa Laval manufactures a comprehensive range of spiral membranes which are widely used for many applications within the food, beverage, dairy, biotech and pharmaceutical industries. The Alfa Laval spiral membranes are available for all cross-flow filtration processes and with different combinations of membrane, diameter, length and spacer. The spiral membranes all feature a full-fit sanitary design
Fine-tuned filtration
Membrane filtration separates out the different components in a feed stream on the basis of the size and shape of the micro-particles within it. And the better the filtration, the better the quality and value of your end product.
The unique design of Alfa Laval spiral membranes makes sure the feed stream passes through the spiral membrane under the best possible flow conditions. The ensures the most efficient separation and superior flux, and that each spiral membrane has a long service life and is easy to clean.
Many applications
Alfa Laval spiral membranes are widely used for many applications within the food, beverage, dairy, biotech and pharmaceutical industies where the aim is to achieve a high-yield concentration and demineralization of products as well as recovery, purification, fractionation etc.
Many combinations
Alfa Laval offers spiral membranes for all cross-flow filtration processes: reverse osmosis, nanofiltration, ultrafiltration and microfiltration.
Our spiral membranes are available with the same type of basic membranes as those found in our range of flat sheet membranes. They are available in different combinations of diameter, length and spacer and they all feature a full-fit sanitary design. The scope includes spiral membranes which are especially resistant to high temperatures and pH-values.
Benefits
- low initial investment and low replacement costs due to long life time
- cost-effective operation due to low energy consumption
- highly compact design requiring minimal floor space and external piping
- excellent chemical and thermal stability with high pH and temperature resistance
- available in different combinations of length, diameter ans spacer size.
- special designs with all materials in compliance with FDA regulations for sanitary applications
- same membrane types available as both flat sheet membranes and spiral membranes
- suitable for an extensive range of applications within the food, dairy, beverage, biotech and pharmaceutical industries
- manufactured by Alfa Laval's own membrane centre
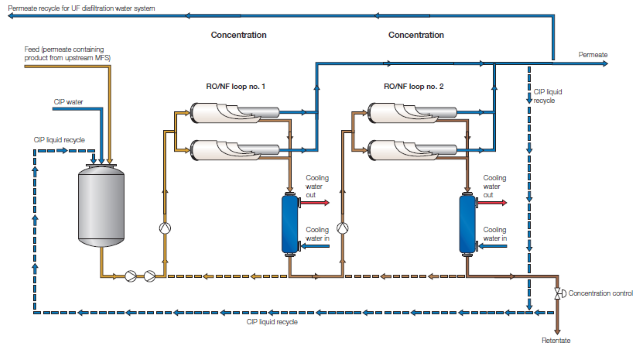
The above illustration shows an Alfa Laval spiral system of the kind typically used for applications such as the concentration of antibiotics, enzymes, yeast extract, egg white and juice as well as for reducing the alcohol content in beer.
The system can also be equipped with ultrafiltration or microfiltration spiral membranes for concentration, clarification and purification.
The spiral concentration process illustrated can also be combined with for example an Alfa Laval spiral or plate-and-frame system for product recovery incorporating diafiltration, which gives the advantage of using permeate from the concentration step as diafiltration water in the recovery process. This significantly reduces the need for process water.

Flow pattern in a spiral membrane from Alfa Laval. Red is feed/retentate. Yellow is permeate
How it works
The core of each spiral element is a perforated central tube, with large membrane pockets attached. Each of these contains a spacer net that transports the permeate out of the membrane pocket and into the central tube.
Different thicknesses of spacer net between each pocket make sure the feed is evenly distributed over the entire surface of the membrane.
The special tight-rolling technique used in Alfa Laval spiral-wound membranes provides the maximum possible active membrane area, which provides correspondingly high permeate capacity. The exceptional firmness characteristics of Alfa Laval spiral-wound membranes ensure long service life and fewer channelling problems, even if a high pressure drop across the element.
Compliances
All materials used for the production of Alfa Laval membranes, in both spiral-wound and flat-sheet membrane designs and configurations, comply with EU Regulation (EC) 1935/2004, EU Regulation 10/2011, EU Regulation (EC) 2023/2006 and FDA regulations (CFR) Title 21. The membranes are thus suitable for use within food and pharmaceutical processing applications.
These compliance also extend to the equipment and fittings related to membrane operations, including items such as plate-and-frame units, element housings and pumps.

